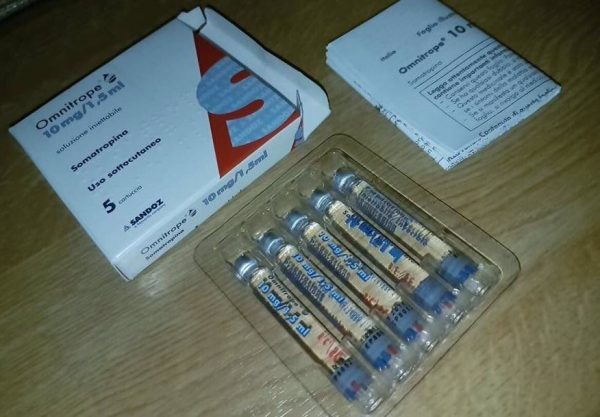
OMNITROPE 10MG/1.5ML – 30IU (30IU x 1 vial) Kit
OMNITROPE 10MG is used to treat growth failure in children and adults who lack natural growth hormone, and in those with chronic kidney failure, Noonan syndrome, Turner syndrome, Prader-Willi syndrome, short stature at birth with no catch-up growth, and other causes. Somatropin is also used to prevent severe weight loss in people with AIDS, or to treat short bowel syndrome.
Buy OMNITROPE 10MG is a polypeptide hormone of recombinant DNA origin. It has 191 amino acid residues and a molecular weight of 22,125 daltons. The amino acid sequence of the product is identical to that of human growth hormone of pituitary origin (somatropin). Omnitrope is synthesized in a strain of Escherichia coli that has been modified by the addition of the gene for human growth hormone.
Side effects OMNITROPE 10MG
Common side effects of OMNITROPE include:
- headache,
- nausea,
- vomiting,
- fatigue,
- muscle pain,
- weakness,
- feeling tired,
- injection site reactions (redness, soreness, swelling, rash, itching, pain, or bruising),
- pain in your arms or legs,
- joint stiffness or pain, or
- cold symptoms such as stuffy nose, sneezing, sore throat.
The dosage and administration schedule should be individualized based on the growth response of each patient, or the condition being treated. Omnitrope may interact with insulin or oral diabetes medicines, steroids, cyclosporine, seizure medication, birth control pills, or hormone replacement medications for men or women. Tell your doctor all medications and supplements you use. Omnitrope should be used only when prescribed during pregnancy. It is unknown if this drug passes into breast milk. Consult your doctor before breastfeeding.
Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in section.
Somatropin must not be used when there is any evidence of activity of a tumor. Intracranial tumors must be inactive and anti-tumor therapy must be completed prior to starting GH therapy. Treatment should be discontinued if there is evidence of tumor growth.
Somatropin must not be used for growth promotion in children with closed epiphyses.
Patients with acute critical illness suffering complications following open heart surgery, abdominal surgery, multiple accidental trauma, acute respiratory failure or similar conditions must not be treated with somatropin (regarding patients undergoing substitution therapy, see section.


Reviews
There are no reviews yet.